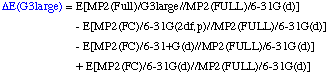G3 Theory
G3 is an improvement over G2 theory in several ways and tries to reproduce effectively QCISD(T)/G3large
energies through a series of calculations at lower level. The G3large basis set is a slightly modified
version of the 6-311+G(3df,2p) basis set used in the G2 procedure. The G3 energy at 0 degrees Kelvin
E0(G3) is defined as:

The definition of the components being:
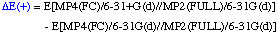
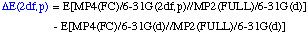
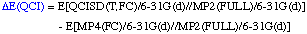
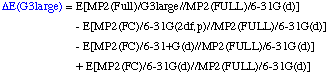

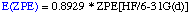
The necessary energies can be calculated most efficiently in the following sequence:
- Optimization and frequency calculation at the HF/6-31G(d) level of theory
- Optimization at the MP2(FULL)/6-31G(d) level of theory
- QCISD(T,FC)/6-31G(d)//MP2(FULL)/6-31G(d) single point
- MP4(FC)/6-31+G(d)//MP2(FULL)/6-31G(d) single point
- MP4(FC)/6-31G(2df,p)//MP2(FULL)/6-31G(d) single point
- MP2(Full)/G3large//MP2(FULL)/6-31G(d) single point
Comments:
- While a 6-311+G(3df) basis set has been used in G2 theory for all first and second row
elements, a different strategy has been pursued in G3 theory. First, a slightly smaller
6-311+G(2df) basis has been chosen for the first row elements, while a larger 6-311+G(3d2f)
basis has been chosen for the second row elements. In both cases core polarization functions
(e.g. p- and d-type for carbon) have also been added. Hydrogen is still treated with a 311+G(2p)
basis. The G3large basis sets can be found on the G3 theory
web site set up by Larry Curtiss.
A local copy can be found here.
- Open shell systems are treated using unrestricted wavefunctions (UHF, UMP2 . . )
- The higher level correction (HLC) is supposed to compensate for the remaining deficiencies
of the method. In contrast to G2 theory, different parameters A and B are used for
atoms and molecules to give the smallest average absolute deviation from experiment.
- Spin orbit correction terms E(SO) (mainly of experimental origin) are added only for atoms
- The mean absolute deviation for the original G2 neutral set (125 reaction energies) is
1.00 kcal/mol. The mean absolute deviation for the extended G2 neutral set (148 reaction
energies) is 0.9 kcal/mol.
Literature:
- L. A. Curtiss, K. Raghavachari, P. C. Redfern, V. Rassolov, J. A. Pople,
"Gaussian-3 (G3) theory for molecules containing first and second-row atoms"
J. Chem. Phys. 1998, 109, 7764 - 7776.
- L. A. Curtiss, K. Raghavachari,
"G2 Theory"
The Encyclopedia of Computational Chemistry, P. v. R. Schleyer (editor-in-chief),
John Wiley & Sons Ltd, Athens, USA, 1998, 2, 1104 - 1114.
- L. A. Curtiss, P. C. Redfern, K. Raghavachari, V. Rassolov, J. A. Pople,
"Gaussian-3 theory using reduced Mřller-Plesset order"
J. Chem. Phys. 1999, 110, 4703 - 4709.
- A. G. Baboul, L. A. Curtiss, P. C. Redfern, K. Raghavachari,
"Gaussian-3 theory using density functional geometries and zero-point energies"
J. Chem. Phys. 1999, 110, 7650 - 7657.
last changes: 01.04.2008, AS
questions & comments to: axel.schulz@uni-rostock.de